History of atomic theory
Adapted from Wikipedia · Adventurer experience
Atomic theory is the scientific idea that everything around us is made of tiny particles called atoms. The word "atom" has changed meaning over time as scientists learned more. At first, people thought atoms were the smallest pieces of matter that could not be broken down any further. Later, scientists discovered that atoms are the building blocks of chemical elements and that they can combine in simple ratios.
This theory is very important in science because it helps us understand how all physical things are made and how they behave. It is essential for many areas of science, including chemistry and physics. In The Feynman Lectures on Physics, famous physicist and Nobel Prize winner Richard Feynman called the atomic idea one of the most powerful thoughts in science.
Philosophical atomism
Main article: Atomism
The idea that everything is made of very small pieces called atoms is very old. It began in ancient times when people thought about how matter works. The word "atom" comes from an ancient Greek word meaning "indivisible." At that time, these were just ideas based on thinking, not on real experiments or science. Today’s science about atoms is very different from these old thoughts.
Pre-atomic chemistry
In the late 1600s, Robert Boyle introduced the idea of a chemical element as a basic substance that is different from a compound. Later, in the late 1700s, Antoine Lavoisier made important discoveries in chemistry. He showed that compounds are made of elements in fixed amounts and that water can be broken down into hydrogen and oxygen, which are elements because they cannot be broken down further. Lavoisier also introduced the law of conservation of mass, which tells us that in a chemical reaction, matter is neither created nor destroyed — the total mass stays the same. Around the same time, Joseph Proust discovered the law of definite proportions, explaining that compounds always break down into elements in the same fixed ratios, no matter how much of the compound there is.
Dalton's chemical atomism
In 1804, John Dalton looked at how elements combine to make compounds. He found that when two elements form more than one compound, the amounts of one element that combine with a fixed amount of the other are in simple whole-number ratios. For example, he studied three compounds made from nitrogen and oxygen and saw that the amounts of oxygen were in the ratios of 1:2:4.
Dalton used these patterns to suggest that all matter is made of tiny, indivisible particles called atoms. He thought that atoms of the same element always have the same weight, while atoms of different elements have different weights. His ideas helped explain how chemicals react and form new substances. Later scientists like Amedeo Avogadro built on Dalton's work to improve his theories.
Opposition to atomic theory
Dalton's atomic theory interested many people, but not everyone agreed with it. One problem was confusion about words. The word “atom” meant something that can’t be divided, but Dalton used it for the smallest piece of any chemical substance. This caused debates about words like “molecule” and “atom.” Over time, scientists made these ideas clearer. Important talks happened at the Karlsruhe Congress in Germany in 1860.
Some scientists also had concerns. Because atoms could not be seen, some thought scientists should only study what they could observe, not guess about things they couldn’t see. These thinkers split into two groups: one group used “equivalent weights” for experiments, and the other thought only energy mattered in physical models. Later discoveries, like the periodic table, helped end these debates.
Prout's hypothesis
Main article: Prout's hypothesis
Early scientists believed that each element was made of unique atoms that all weighed the same. In 1815, a scientist named William Prout proposed that all matter might be made of tiny pieces of one basic element. He called this basic piece a protyle and linked it to hydrogen. Another scientist, Berzelius, disagreed because measurements at the time did not support this idea. Later, in 1912, Francis Aston discovered some evidence that supported part of Prout’s idea.
Vortex theory
Main article: Vortex theory of the atom
From the 1860s to around 1890, some scientists thought atoms might be like spinning rings of fluid. They imagined matter as stable swirls in an invisible fluid, similar to smoke rings we can see. This idea was inspired by math related to fluid movement but didn’t fully explain things like magnetism or gravity. Even its supporters later found the idea didn’t work well.
Isomerism
Scientists found that some substances can have the same chemical ingredients but behave differently. In 1827, Friedrich Wöhler showed that silver fulminate and silver cyanate have the same amounts of silver, carbon, nitrogen, and oxygen, but their properties are different. In 1830, Jöns Jacob Berzelius named this phenomenon isomerism.
Later, in 1874, Jacobus Henricus van 't Hoff suggested that atoms in molecules arrange themselves in specific shapes. His idea helped explain why some compounds can have different structures while using the same atoms. For example, the molecule pentane (C5H12) has three possible arrangements. These are called n-pentane, isopentane, and neopentane.
Mendeleev's periodic table
Main article: Periodic table
In 1869, Dmitrii Mendeleev noticed that when elements were arranged by their atomic weights, they showed repeating patterns in their properties. For example, lithium, sodium, and potassium shared similar traits. Using these patterns, Mendeleev predicted elements that had not yet been discovered, such as scandium, gallium, and germanium.
The original periodic table was arranged by atomic weight, but sometimes elements were swapped to group those with similar properties together. For example, tellurium is placed before iodine so iodine can be in the same column as other halogens. Today, the periodic table is ordered by atomic number. When Mendeleev created his table, one entire row was missing because the noble gases had not yet been found.
Kinetic theory of gases
Main article: Kinetic theory of gases
In 1738, Daniel Bernoulli said that the pressure of gases and heat come from tiny particles called molecules moving. He used this to explain a rule about gases, called the ideal gas law. Later, in the 1860s, James Clerk Maxwell brought back this idea and said that gas particles move at different speeds. Around the same time, Ludwig Boltzmann used these ideas to explain how heat and energy work together. In 1905, Albert Einstein used these ideas to explain why tiny particles in water move around randomly. This was first seen by Robert Brown in 1827 and tested later by Jean Perrin in 1908.
| Molecule | Perrin's 1909 measurements: 50 | Modern measurements |
|---|---|---|
| Helium | 1.7 × 10−10 m | 2.6 × 10−10 m |
| Argon | 2.7 × 10−10 m | 3.4 × 10−10 m |
| Mercury | 2.8 × 10−10 m | 3 × 10−10 m |
| Hydrogen | 2 × 10−10 m | 2.89 × 10−10 m |
| Oxygen | 2.6 × 10−10 m | 3.46 × 10−10 m |
| Nitrogen | 2.7 × 10−10 m | 3.64 × 10−10 m |
| Chlorine | 4 × 10−10 m | 3.20 × 10−10 m |
Plum pudding model
Main articles: Electron and Plum pudding model
Before 1899, people thought atoms were the smallest pieces of matter. Then, J. J. Thomson discovered the electron using special glass tubes called Crookes tubes. These tubes have two electrodes with a vacuum between them. When voltage is applied, glowing rays called cathode rays appear. Thomson found these rays were made of tiny, negatively charged particles. He called them "corpuscles." He measured their mass and charge and found they were much smaller than atoms.
In 1904, Thomson suggested a new model for atoms. He thought the atom was like a sphere filled with positive charge, with tiny electrons scattered inside, like raisins in a pudding—hence the name "plum pudding model." This model tried to explain how atoms could be neutral overall, with both positive and negative charges balanced inside.
Planetary models
In the late 1800s, scientists imagined how atoms might be built. They thought electrons moved around like planets.
In 1903, a scientist named Hantaro Nagaoka suggested a new idea. He said electrons circled a big, positive center, like moons around Saturn. This idea got people talking, but it didn’t explain everything about atoms. Nagaoka explored other theories after 1908.
Haas atomic model
In 1910, Arthur Erich Haas had a new idea about atoms. He thought that in a hydrogen atom, an electron moves around a sphere of positive charge.
Haas used math to show how the forces work together. His ideas helped other scientists learn more about atoms. Later, a scientist named Bohr used similar ideas to find the size of the hydrogen atom. This size is called the Bohr radius.
Nicholson atom theory
In 1911, John William Nicholson made a model of the atom. In this model, tiny parts called electrons moved around a center part called a nucleus. This was like another model made by J.J. Thomson called the plum pudding model. Nicholson looked at light from stars and planets to help create his ideas.
Nicholson's model is not talked about much today, but it was helpful for later work. His ideas helped another scientist, Niels Bohr, think about atoms. Bohr later showed that hydrogen atoms had just one electron, not two as Nicholson thought.
Discovery of the nucleus
Main article: Rutherford scattering experiments
In 1911, a scientist named Ernest Rutherford changed what we thought about atoms. He and his team shot tiny, positive particles called alpha particles at thin metal sheets. Some of these particles bounced back at big angles, which was surprising.
Rutherford discovered that atoms have a tiny, dense center where most of the weight and positive charge is located. This center is called the atomic nucleus. His discovery helped scientists learn how atoms are built. Later, a scientist named Niels Bohr added ideas about how electrons move around this nucleus.
Discovery of isotopes
Main article: Isotope
At the same time that scientists like Rutherford were studying atoms, a chemist named Frederick Soddy was looking at radioactive materials. He found that some elements that acted the same in experiments were actually different inside. He called these isotopes. Later, a scientist named Francis Aston built a tool to measure these small differences. He learned that each isotope had a whole number mass, except for hydrogen. His work helped us learn more about the tiny pieces that make up everything.
Atomic number
Before 1913, scientists thought the properties of elements depended on their atomic weight. But this didn’t always match what was seen in the periodic table, like with cobalt and nickel.
In 1913, Antonius van den Broek suggested that elements should be organized by their charge, called Z, instead of atomic mass. This idea helped clear up some problems in the periodic table.
Later, Moseley tested this idea using X-ray spectroscopy. He found that a special type of X-ray from each element, the K-alpha line, was linked to the element’s charge, or atomic number, Z. His work showed a clear link between X-ray frequencies and the atomic numbers of many elements.
Bohr model
Main article: Bohr model
In 1913, Niels Bohr created a new way to understand how atoms work. He thought electrons could only move between certain energy levels. This helped explain patterns in light from atoms, especially for hydrogen. Bohr's model was simple and worked best for hydrogen, but it was an important step in learning about atoms. Later, scientists built on his ideas to understand more complex atoms.
Discovery of the proton
In 1815, William Prout noticed that the weights of atoms seemed to be multiples of hydrogen’s weight. This suggested that all atoms might be made of hydrogen. But this idea had problems until isotopes were found in 1913.
Later, in 1917, Rutherford shot alpha particles at nitrogen gas. He saw hydrogen ions come off. This helped him discover the proton, a tiny part inside atoms with a positive charge. The number of protons in an atom’s nucleus is called the atomic number. This number helps us tell different chemical elements apart.
Quantum mechanical models
Main article: History of quantum mechanics
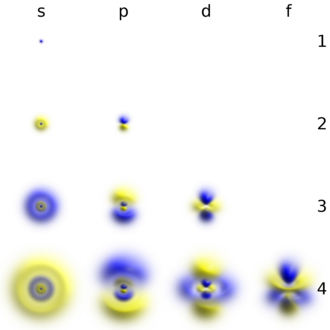
In 1924, Louis de Broglie proposed that tiny particles like electrons can act like waves. Erwin Schrödinger used this idea to make a math model of electrons as waves, called a wave function. This helped solve problems that older models couldn't.
Schrödinger’s model showed electrons in areas named atomic orbitals. These orbitals have different shapes based on energy and angular momentum. These shapes help explain how atoms join to form molecules. Electrons fill these orbitals following a rule known as the Pauli principle, which sets a limit on how many electrons can be in each orbital.
Discovery of the neutron
Main article: Discovery of the neutron
In the 1920s, scientists thought atoms had tiny particles inside called "nuclear electrons." They believed these helped explain some properties of elements. In 1928, Walter Bothe found that when beryllium was hit with special particles, it gave off a strange kind of radiation. At first, people thought this was gamma radiation. But later, James Chadwick showed that this radiation was actually made of tiny, neutral particles with mass similar to protons. He named these particles "neutrons." This discovery changed how we understand atoms.
Images
Related articles
This article is a child-friendly adaptation of the Wikipedia article on History of atomic theory, available under CC BY-SA 4.0.
Images from Wikimedia Commons. Tap any image to view credits and license.
 Safekipedia
Safekipedia